Regulatory
Accelerating regulatory success across the product lifecycle - from first submission to global compliance.
Driving regulatory excellence
Tailored solutions and data-driven insights that position your organisation to excel in a competitive market.
End-to-end regulatory support
From regulatory strategy through submission, approval and ongoing lifecycle management — one partner across the entire journey. This positions BASE against fragmented vendor landscapes where clients juggle multiple providers.
Our experienced team improves operational efficiency and strengthens compliance through regulatory expertise.
Future-ready regulatory operations
GenAI-driven solutions, IDMP readiness and adaptable frameworks designed to evolve with the regulatory landscape. Stay ahead of shifting requirements while building operations that scale with your portfolio and ambitions.

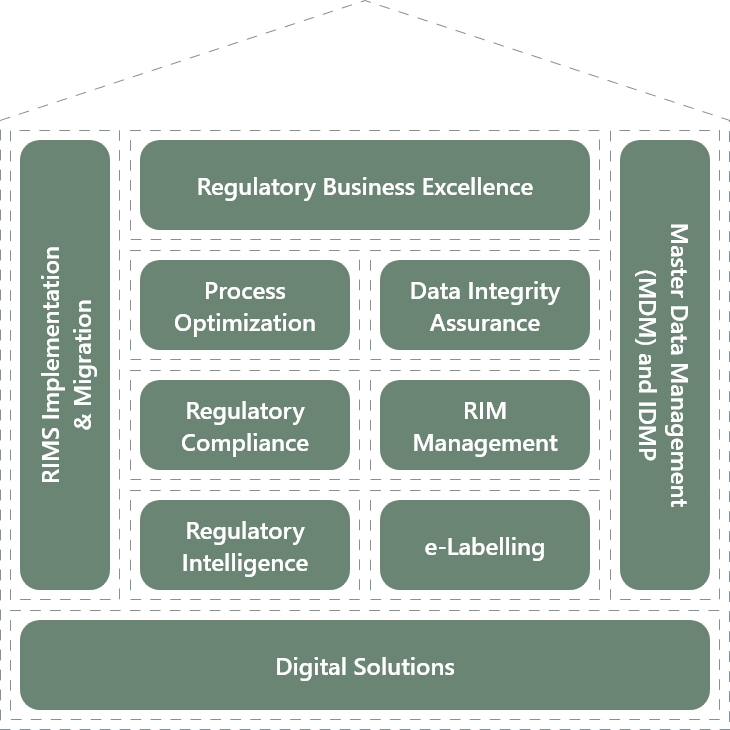
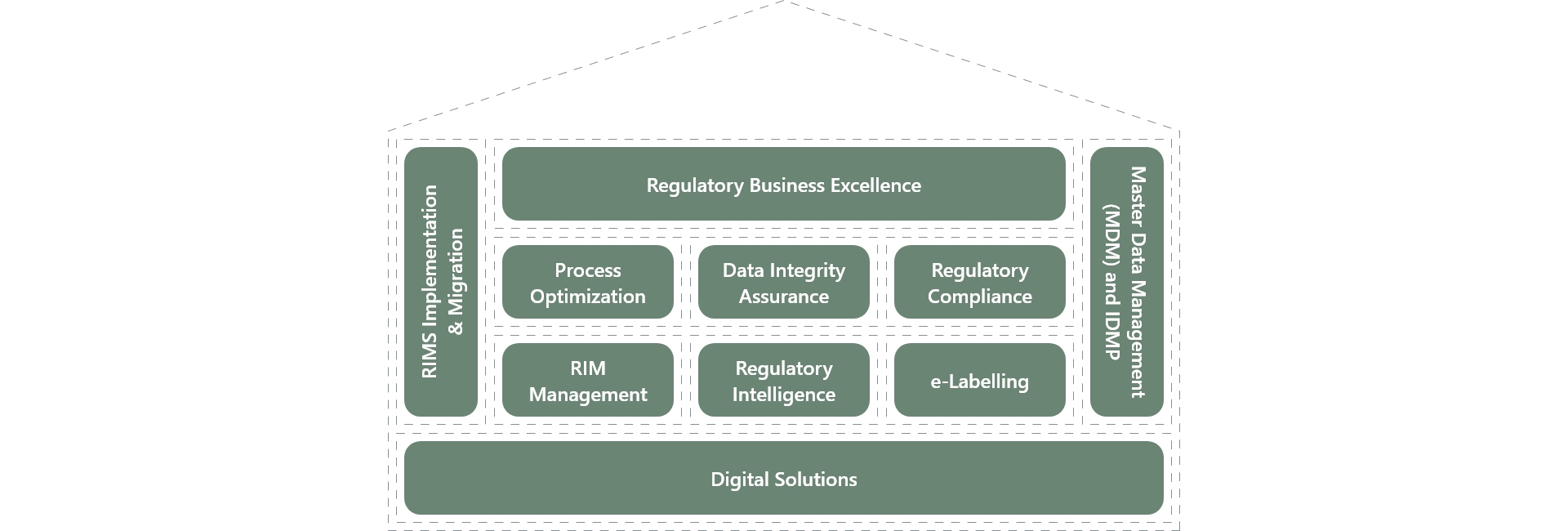
Our Offerings
Advisory & Consulting
Navigating the regulatory landscape requires more than compliance, it demands strategic clarity. Our advisory services help life sciences organisations assess where they stand, identify what needs to change and build a clear path forward.
Maturity assessment
Understanding where your regulatory operations stand is the first step to improving them. We evaluate your processes, data and technology against industry benchmarks to identify gaps, highlight strengths and provide a clear roadmap that keeps you inspection-ready and aligned with compliance requirements.
Vendor selection
Choosing the right technology and service providers can shape your regulatory operations for years. We support the full selection journey — from market research and vendor evaluation through due diligence and negotiation — so you make informed decisions that drive long-term operational efficiency.
Process mapping, design and optimisation
Inefficient workflows and disconnected systems create compliance risk and slow your teams down. Through in-depth analysis of your processes and IT landscape, we identify gaps and design tailored solutions that improve data flows, system integration and overall operational impact.
Regulatory intelligence advisory
We track regulatory trends, interpret legislative changes and assess their impact on business operations. Our insights help pharmaceutical companies maintain compliance, optimise product development pathways and stay ahead in a constantly evolving regulatory landscape.
AI & Automation
Manual processes slow down submissions, increase error rates and drain expert resources. Our AI and automation solutions remove these bottlenecks, accelerating timelines, improving accuracy and freeing your teams to focus on high-value regulatory work.
Health Authority Query Management agent
Health authority queries demand fast, accurate responses under tight deadlines. Our HAQ solution automates response preparation by extracting relevant information from your regulatory documentation, drafting responses and tracking timelines. Teams spend less time searching and compiling, and more time reviewing and finalising — reducing response times while maintaining the quality health authorities expect.
Submission Package Generation & QC
Assembling submission packages is complex, repetitive and error-prone. Our solution automates package generation and runs comprehensive quality checks against EU and US regulations — detecting errors, mismatches and missing details before anything reaches a health authority. With real-time annotations and feedback, your teams can focus on critical submission requirements rather than manual verification.
Global Label Management
Managing labels across global markets means navigating multiple languages, formats and regulatory requirements simultaneously. Our labelling solution streamlines label creation, management and translation across 130+ languages while preserving original document formatting. Built-in detection of inconsistencies and spelling errors ensures accuracy and label consistency across markets, simplifying global submissions
Automated Audit Log
Regulatory compliance depends on visibility into who did what and when. Our Automated Audit Log continuously monitors Vault activities, flags unusual behaviour and enables workflow triggers to safeguard regulatory information. Running as a daily automated process, it replaces manual audit log reviews, improving efficiency, strengthening security and keeping your compliance posture intact.


Data & Insights
Reliable regulatory operations start with trusted data. Our data governance and insights services help organisations take control of their regulatory information — standardising, structuring and enriching it to meet both current requirements and future expectations.
Master Data Management (MDM) Governance
Our framework provides structured oversight and control of critical regulatory data. The framework consolidates, standardises and synchronises information across your organisation to establish a single source of truth. This approach strengthens compliance tracking, improves data retrieval and streamlines regulatory submissions, supporting informed decision-making and operational efficiency.
IDMP journey
Our IDMP offering supports the standardisation of medicinal product definitions to enable clear identification and information exchange, particularly for adverse events. Through application of advanced technologies, we assess data sources, enrich quality and fully implement IDMP requirements, aligning business processes with regulatory expectations.
e-Product Information (ePI)
Our ePI service helps clients digitise and manage product information, including SmPCs, package leaflets and labelling, ensuring secure and compliant distribution across web and digital platforms. This approach streamlines access to authorised medicinal product details while supporting regulatory compliance.
Data Migration
Data Cleansing & Enrichment
Successful migration starts with clean data. Our data cleansing services identify duplicates, inconsistencies, outdated records and metadata gaps across your regulatory information. We then enrich and standardise the data to align with your target system’s requirements — reducing migration risk, shortening timelines and ensuring your new environment starts with a foundation you can trust
Regulatory data migrations carry significant risk, data integrity issues, metadata gaps and system incompatibilities can derail timelines and compromise compliance. Our BASE Migration Framework, built on established QMS and software guidelines, is designed to mitigate these risks. With refined mapping rules, flexible deployment across cloud environments (AWS, Azure, GCP), and agile delivery, we provide continuous transparency and high-quality outcomes across Veeva Vault and custom in-house systems.
Data Integrity Assurance
We uphold data authenticity, traceability and reliability throughout its lifecycle to maintain integrity in highly regulated pharmaceutical environments. Covering data creation, maintenance and disposal, our service mitigates risks of errors and manipulation, preserving data quality for confident decision-making.

End-to-end Implementation
As part of our Veeva Value Journey, we provide comprehensive support for deploying the Veeva Vault RIM solution, including:
- Vault Registrations – Streamlining product registration and lifecycle management.
- Vault Submissions – Managing regulatory submissions efficiently.
- Vault Submissions Archive – Maintaining a structured, compliant repository of regulatory submissions.
- Vault Submissions Publishing – Automating content preparation and submission publishing.
- Vault Connections – Enabling smooth data exchange across regulatory systems.
We provide comprehensive support, covering system configuration, data mapping, migration, user training and ongoing assistance.
Our Veeva-certified consultants collaborate with your team to assess and align existing processes and regulatory data with Veeva Vault RIM, enabling a smooth transition while strengthening data integrity and improving operational efficiency.
Managed Services
Veeva Vault RIM management (L1/L2 support)
Our Veeva Vault RIM management service provides dedicated Level 1 and Level 2 support, helping organisations meet global regulatory requirements with confidence. Through comprehensive managed services, we reduce administrative burden, streamline regulatory workflows, uphold compliance and accelerate time to market.

The regulatory house of excellence